Caffeine, Calcium and Catch: Investigating the Effect of Caffeine on Calcium Dynamics in the Mussel Catch State
Experimental Write-Up Assignment for Principles of Animal Physiology (BIO 329L) taught by Dr. Sheila Patek and Dr. Emily Ozdowski at Duke University
October 2025In bivalve mussels (Mytilus), the catch state is a unique physiological condition that allows its smooth muscle to maintain prolonged contraction and shell closure with minimal energy use. In lecture, we emphasized the difference between typical vertebrate muscle contractions, which requires continuous ATP hydrolysis to sustain force, and the mechanism underlying catch, which rely on Twitchin, a thick filament–associated protein that can bind to actin when dephosphorylated, stabilizing cross-bridges between actin and myosin even after cytosolic calcium levels decline, allowing the muscle to remain tense without further calcium cycling or ATP consumption. The initiation and maintenance of the catch state depends on precise calcium ion (Ca²⁺) regulation mediated by ryanodine receptor (RyR) channels on the sarcoplasmic reticulum (SR). RyRs open to release Ca²⁺ into the cytosol during contraction and close as Ca²⁺ is re-sequestered for relaxation (Patek, 2016).
Caffeine is a known agonist of RyR channels in vertebrate skeletal and smooth muscles, binding to the receptor and stabilizing its open conformation (Shi et al. 2003, Kong et al. 2008). Intracellular Ca²⁺ levels play a crucial role in initiation of the catch state, with dephosphorylation of twitchin occurring once intracellular Ca²⁺ concentration returns to below a certain threshold, establishing and maintaining catch (Sugi et al. 2020). We hypothesized that, similar to the well-studied mechanisms in vertebrates, caffeine will bind to RyR channels, stabilizing its open conformation, and leading to persistent leakage even below normal activation thresholds. This disrupts the normal transition into and out of the catch state by prolonging Ca²⁺ availability and delaying relaxation, reflected in a decreased rate of relaxation.
The present experiment tested this hypothesis by measuring the rate of relaxation of dissected byssal muscles given a low (0.005mM) and high (0.5mM) dose of caffeine compared to controls bathed in Instant Ocean artificial seawater. All procedures and baseline calibrations followed the standardized setup described in Patek (2016). Each pre-weighed mussel received 1mL of Serotonin (5HT) and was given time to relax from any tension built during dissection and set-up of the force transducer apparatus. When a baseline voltage was achieved, the mussel received its treatment (Control N=2, Low N=2, High N=2). 5 minutes after the treatment, the mussel was given its first 1mL dose of Acetylcholine (ACh) and the resulting contraction amplitude (V), latency to contract (s), time of contraction and time to relaxation (s) were recorded. A maximum of 2 additional 1mL doses of ACh were given to mussels exhibiting activity, each spaced 5 minutes apart from the first dose.
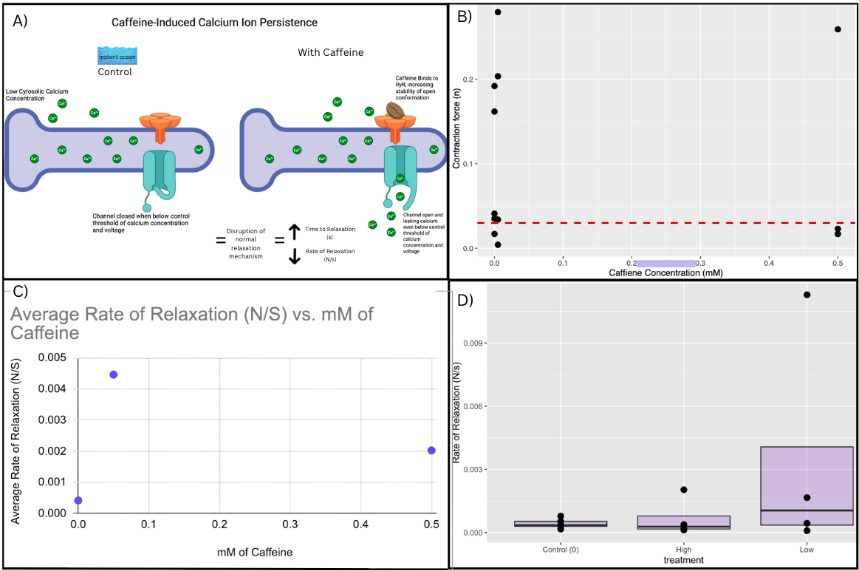
Contraction Amplitude (V) was converted to Force (N) using the standard acceleration due to gravity 9.81 m/s2. Out of all recorded contractions (N=13), 8 were above the contraction force threshold of 0.030N. This threshold was developed in order to calculate the average rate of relaxation (N/s), and following instructor input on minimum voltage amplitude for a reliable muscle contraction. The average rate of relaxation for each treatment was calculated as the mean of the individual relaxation rates obtained from each reliable muscle contraction, where each rate represented the change in force (ΔN) over the change in time (Δt) during the relaxation phase.
Across all trials, there was no significant relationship between caffeine concentration and contraction force or relaxation rate in mussel catch muscle. The average rate of relaxation was 0.0042N/s for control treatments, 0.0045N/s for low caffeine doses, and 0.0020N/s for high caffeine doses, displaying no clear directional trend. The scatter plot of contraction force versus caffeine concentration also showed no relationship, even when considering the developed 0.030N threshold, and the boxplots of relaxation rate revealed broadly overlapping distributions among all three treatment groups. Thus, although caffeine is known to act as an RyR agonist in vertebrate muscle, the data collected here did not support our prediction in bivalve mussels, showing no significant correlation or directional shift across treatments.
Figure 1: Caffeine, a RyR agonist for vertebrate muscles, did not have a significant effect on rate of relaxation at low (0.005 mM) and high (0.5 mM) doses in invertebrate bivalve mussels. A) Conceptual model of the mechanistic hypothesis: caffeine binds RyR channels, stabilizing its open conformation and maintaining partial calcium leakage even below normal threshold levels. This disrupts relaxation by prolonging cytosolic Ca²⁺ presence, delaying the dephosphorylation of Twitchin. Created with Biorender and modified in Canva. B) Scatter plot of average contraction force vs. caffeine concentration shows large variation, resulting in the development of a significance threshold of 0.030N for contraction force. Contractions below this threshold were not included in calculation of average rates of relaxation. C) Scatterplot of caffeine concentration (mM) vs. average rate of relaxation (N/s) shows no significant relationship. D) Boxplot comparison shows no significant relationship between treatment and rate of relaxation (N/s).
AI Assessment Statement
I used Duke’s licensed ChatGPT to help consolidate and refine this report for clarity and conciseness. For example, similar to Build 1, I prompted it: “what are the scientifically essential elements of this sentence and what can be changed when there is a page limit?” It was helpful in breaking down complex ideas and suggesting where content was essential to the meaning of the sentence and what was “fluff”, but less helpful in helping to reconstruct sentences. I believe I used AI ethically and ensured all interpretations and final wording reflected my own understanding of the material.
References
Kong, H., Jones, P. P., Koop, A., Zhang, L., Duff, H. J., & Chen, S. R. (2008). Caffeine induces Ca2+ release by reducing the threshold for luminal Ca2+ activation of the ryanodine receptor. The Biochemical journal, 414(3), 441–452. https://doi.org/10.1042/BJ20080489
Patek, S. (2016). Adaptation of Mytilus Anterior Byssal Retractor Muscle. iWorx Animal Physiology Lab Manuals: Animal Muscle (AM08), 1-19.
Shi, D., Padgett, W. L., & Daly, J. W. (2003). Caffeine analogs: effects on ryanodine-sensitive calcium-release channels and GABAA receptors. Cellular and molecular neurobiology, 23(3), 331–347. https://doi.org/10.1023/a:1023688604792
Sugi, H., Ohno, T., & Moriya, M. (2020). Mechanism and Function of the Catch State in Molluscan Smooth Muscle: A Historical Perspective. International journal of molecular sciences, 21(20), 7576. https://doi.org/10.3390/ijms21207576