Testing Physiological Demand: Exercise, Feeding and Blood Glucose Dynamics in Zebrafish
Experimental Write-Up Assignment for Principles of Animal Physiology (BIO 329L) taught by Dr. Sheila Patek and Dr. Emily Ozdowski at Duke University
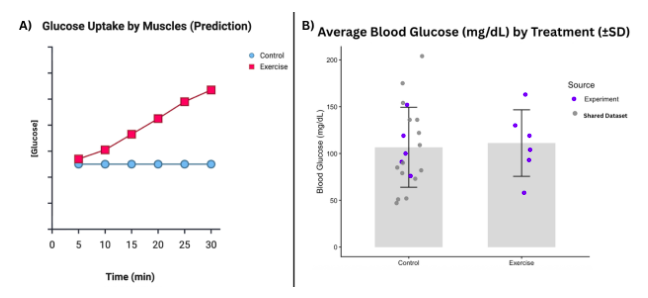
November 2025This experiment tested whether brief post-feeding exercise decreases circulating blood glucose in zebrafish by increasing muscle glucose uptake. We hypothesized exercised zebrafish would have lower blood glucose than controls at 30 minutes postprandial. Following an 18-hour fast and standardized feeding, zebrafish were assigned to exercise (N=6, 30-minute mild current) or control groups (N=21, 6 in still water from current experiment + 15 shared data set values), with blood glucose measured immediately after treatment (Coppola, 2016). The results contradicted our hypothesis: controls averaged 107.6 mg/dL (range: 50-200) while exercised fish averaged 111.2 mg/dL (range: 60-160), with overlapping standard deviations indicating no significant exercise effect on circulating glucose.
The absence of reduced blood glucose in exercised zebrafish suggests that 30 minutes of low-intensity swimming was insufficient to produce measurable glucose clearance from circulation. Several physiological mechanisms may explain this finding. First, exercise-induced glucose uptake depends on GLUT4 transporter translocation, which requires adequate metabolic demand (Ren et al., 1994). The mild current generated by our magnetic stir bar may not have created sufficient energetic stress to trigger substantial GLUT4 activation beyond basal levels. Second, at 30 minutes post-feeding, active glucose absorption from the digestive tract likely continued, potentially masking any exercise-induced uptake by muscle (Seiiliez et al., 2013; Handeland et al., 2008; Navarro-Guillen & Yufera, 2025). If absorption rates match or exceed muscle uptake rates, circulating glucose remains stable despite insulin-stimulated GLUT4 translocation (Shepherd & Khan, 1999; James et al., 1988; Gromova et al., 2021). Third, zebrafish possess homeostatic mechanisms that tightly regulate blood glucose through compensatory processes such as hepatic glucose release, which may maintain circulating concentrations despite increased muscle demand (Hill et al., 2022; Gromova et al., 2021; Seiliez et al., 2013). These results align with studies in mammals showing that low-intensity exercise produces minimal acute changes in blood glucose compared to moderate or high-intensity activity (Adams, 2013; Marliss & Vranic, 2002; Wahren et al., 1978; Chen et al., 2003), suggesting that exercise intensity thresholds for measurable metabolic effects may be conserved across vertebrates.
Thus, a critical limitation was the lack of quantified exercise intensity. The magnetic stir bar generated an unmeasured current that may have been insufficient to elicit significant metabolic demand (Ren et al., 1994; Marliss et al., 200). Without measuring swimming speed or current velocity, we cannot confirm our fish experienced true aerobic exercise versus minimal effort to maintain position. This limitation likely shifted both groups toward similar metabolic states, contributing to overlapping glucose values. Future studies should use flow chambers with submersible pumps and flow meters to generate verified current speeds that match natural zebrafish swimming conditions (3.5-13.9 cm/s) and are known to elicit measurable metabolic responses (Arunachalam et al., 2013; Mwaffo et al., 2017; Illling et al., 2021).
Additionally, this experiment assumed GLUT4 translocation and glucose uptake occur rapidly enough to affect circulating levels within 30 minutes. Studies show that peak postprandial glucose occurs up to 60 minutes after feeding when absorption rates are highest (Furuichi & Yone, 1981; Eames et al., 2010; Martins et al., 2024). Thus, our 30-minute measurement likely captured ongoing glucose absorption that would elevate values in both groups and obscure any exercise-induced decrease in circulating glucose.
Future research should compare glucose dynamics across a range of exercise intensities and frequencies to help establish intensity thresholds for metabolic effects (Marliss et al., 2002; Chen et al., 2003; Adams, 2013). Additionally, measuring stress hormones across the exercise period could distinguish between stress responses and exercise-mediated changes in glucose homeostasis (Martins et al., 2024; Hill et al., 2022)
This study contributes to our understanding of glucose regulation in zebrafish by demonstrating that brief, low-intensity exercise does not produce acute changes in circulating glucose, highlighting the importance of considering exercise intensity, timing, and homeostatic mechanisms when using zebrafish as a metabolic model for vertebrate physiology.
Figure: The results did not support the hypothesis that blood glucose levels decrease for zebrafish exercised for 30 minutes in comparison to controls. A) Mechanistic Hypothesis: Exercise will decrease blood glucose after 30 minutes from feeding by increasing [Glucose] transported to the muscles (glucose uptake by muscles) within 30 minutes from feeding. B) Bar graph of results displaying average blood glucose (mg/dL) after 30 minutes for control and exercise treatment groups. Average values are overlaid with standard deviation bars and dataset values, color coded by source: from the current experiment or from the shared dataset.
Acknowledgements
This analysis included 15 values from the Zebrafish Shared Dataset Fall 2025. Values were collected from two groups of students: Rachel, Savannah, Haley, Anne (Rows 59-61) and Cat, Hailey, Kennedy, Lydia (Rows 432-434, 460-462), as well as from Dr. O (Rows 86-90, 245-247, 432-434, 460-462).
AI Assessment Statement
In preparing this paper, I used Duke’s licensed ChatGPT Edu, to help me structure and refine my discussion. The tool helped me to create an outline ensuring I address all required appointments and organize my thoughts before drafting. I also used the tool to condense overly detailed sentences while maintaining scientific accuracy. For example, I asked the tool to make a sentence I wrote more concise so I could include it without going over the page limit. The tool was helpful in guiding the order of information presented in my discussion and in breaking down complex sentences into more concise descriptions without losing meaning. The tool was most helpful in maintaining conciseness to fit the 2-page limit while preserving depth of analysis.
References
Adams, O. P. (2013). The impact of brief high-intensity exercise on blood glucose levels. Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy, 6, 113–122. https://doi.org/10.2147/DMSO.S29222
Arunachalam, M., Raja, M., Vijayakumar, C., Malaiammal, P., & Mayden, R. L. (2013). Natural history of zebrafish (Danio rerio) in India. Zebrafish, 10(1), 1–14. https://doi.org/10.1089/zeb.2012.0803
Chen, Z.-P., Stephens, T. J., Murthy, S., Canny, B. J., Hargreaves, M., Witters, L. A., Kemp, B. E., & McConell, G. K. (2003). Effect of exercise intensity on skeletal muscle AMPK signaling in humans. Diabetes, 52(9), 2205–2212. https://doi.org/10.2337/diabetes.52.9.2205
Coppola, A. (2016). Measuring blood glucose in adult zebrafish. Animal Physiology Lab Manuals: Metabolism Regulation in Zebrafish, 1, 8–9.
Eames, S. C., Philipson, L. H., Prince, V. E., & Kinkel, M. D. (2010). Blood sugar measurement in zebrafish reveals dynamics of glucose homeostasis. Zebrafish, 7(2), 205–213. https://doi.org/10.1089/zeb.2009.0640
Furuichi, M., & Yone, Y. (1981). The utilization of carbohydrates by fishes. 3. Change of blood-sugar and plasma-insulin levels of fishes in glucose-tolerance test. Bulletin of the Japanese Society of Scientific Fisheries, 47(6), 761–764.
Gromova, L. V., Fetissov, S. O., & Gruzdkov, A. A. (2021). Mechanisms of glucose absorption in the small intestine in health and metabolic diseases and their role in appetite regulation. Nutrients, 13(7), 2474. https://doi.org/10.3390/nu13072474
Hill, R. W., Cavanaugh, D. J., & Anderson, M. (2022a). Chapter 1: Animals and environments. In Animal Physiology (5th ed., pp. 3–32). Oxford University Press.
Hill, R. W., Cavanaugh, D. J., & Anderson, M. (2022b). Chapter 16: Endocrine and neuroendocrine physiology. In Animal Physiology (5th ed., pp. 441–476). Oxford University Press.
Illing, B., Severati, A., Hochen, J., Boyd, P., Raison, P., Mather, R., Downie, A. T., Rummer, J. L., Kroon, F. J., & Humphrey, C. (2021). Automated flow control of a multi-lane swimming chamber for small fishes indicates species-specific sensitivity to experimental protocols. Conservation Physiology, 9(1). https://doi.org/10.1093/conphys/coaa131
James, D. E., Brown, R., Navarro, J., & Pilch, P. F. (1988). Insulin-regulatable tissues express a unique insulin-sensitive glucose transport protein. Nature, 333(6169), 183–185. https://doi.org/10.1038/333183a0
Marliss, E. B., & Vranic, M. (2002). Intense exercise has unique effects on both insulin release and its roles in glucoregulation: Implications for diabetes. Diabetes, 51(Suppl. 1), S271–S283. https://doi.org/10.2337/diabetes.51.2007.s271
Martins, M. S., Carneiro, W. F., Monteiro, K. S., Dos Santos, A. C., Silva, E. P., Luz, R. K., & Fortes-Silva, R. (2024). Metabolic effects of physical exercise on zebrafish (Danio rerio) fed a high-fat diet. Journal of Comparative Physiology B, 194, 793–804. https://doi.org/10.1007/s00360-024-01577-x
Mwaffo, V., Zhang, P., Romero Cruz, S., & Porfiri, M. (2017). Zebrafish swimming in the flow: A particle image velocimetry study. PeerJ, 5, e4041. https://doi.org/10.7717/peerj.4041
Navarro-Guillén, C., & Yúfera, M. (2025). Understanding rhythms in the digestive functionality of fish gut. Journal of Experimental Biology, 228(14), jeb249942. https://doi.org/10.1242/jeb.249942
Ren, J. M., Semenkovich, C. F., Gulve, E. A., Gao, J., & Holloszy, J. O. (1994). Exercise induces rapid increases in GLUT4 expression, glucose transport capacity, and insulin-stimulated glycogen storage in muscle. The Journal of Biological Chemistry, 269(20), 14396–14401.
Seiliez, I., Médale, F., Aguirre, P., Larquier, M., Lanneretonne, L., Alami-Durante, H., Panserat, S., & Skiba-Cassy, S. (2013). Postprandial regulation of growth- and metabolism-related factors in zebrafish. Zebrafish, 10(2), 237–248. https://doi.org/10.1089/zeb.2012.0835
Shepherd, P. R., & Kahn, B. B. (1999). Glucose transporters and insulin action: Implications for insulin resistance and diabetes mellitus. The New England Journal of Medicine, 341(4), 248–257. https://doi.org/10.1056/NEJM199907223410406
Wahren, J., Felig, P., & Hagenfeldt, L. (1978). Physical exercise and fuel homeostasis in diabetes mellitus. Diabetologia, 14, 213–222. https://doi.org/10.1007/BF01219419